 Oxyacids occur when chlorine reacts with alkaline solutions.Although its primary oxidation number is a negative one, chlorine can on rare occasions exhibit plus one, +3, +4, +5, +6, and +7.The first-ever chain chemical reaction was observed by German chemist Max Bodenstein in 1913 when he made chlorine and hydrogen gas explode upon light exposure.In organic chemical reaction mechanisms, chlorine often replaces hydrogen atoms within molecules.Chlorine is the 21st most abundant element in the earth’s crust. 
Gaseous chlorine is so pungent most humans can detect the smell of it in concentrations as low as 3.5 ppm.Chlorine is the third most abundant element in the earth’s oceans.Chlorine is the ninth most abundant element in the human body.As one of the diatomic elements, along with hydrogen, fluorine, iodine, bromine, oxygen, and nitrogen, lone chlorine atoms seldom exist in nature.Chlorine has a storied history of its use for atrocities in war but it also has numerous applications and uses in our modern world from food to consumer products to the environment. Chlorine typically exists in nature as a diatomic element but the single species Cl- is essential to many compounds. It gets its name from the Greek word Kloros, meaning greenish-yellow. It belongs to the halogen family of elements, along with fluorine and bromine. 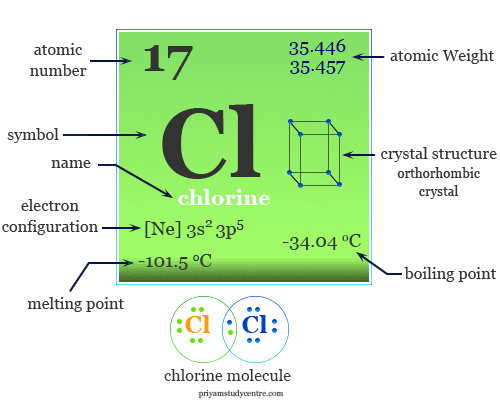
The element chlorine of the periodic table is a clear yellowish-green gas. The Element Chlorine Introduction to Chlorine 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
